[ Silanol compound : RnSi(OH)4n ] + Solvent
Si(OC2H5)4), water (H2O) , isopropanol ((CH5)2CH(OH)) and hydrochloric
acid (HCl).![]()
Because TEOS and water are immiscible, the reactions
only begin when the solvent
isopropanol is added. The reactions took place at 82oC, the boiling
temperature of
isopropanol. The reaction rate can be controlled by adding an acid as
catalyst;
hydrochloric acid was used in this case.
![]()
even with excess water (r >> 2), the reaction does not
go to completion.
Instead, a spectrum of intermediate species are generated. The
intermediates, which
remain soluble in the alcohol-water medium, are silanols, ethoxylanols,
and
polysiloxanes. Depending on this molar ratio, different kinds of
product, e.g. bulk gel,
film, fiber, and power, could be obtained. Beside the r value, the
catalyst type and
concentration, the solvent, temperature, and pressure also cause
modifications in the
structure and properties of the polysilicate products.
The final product (silicate SOG) will be a transparent
sol of organic solvents (ethanol,
acetone, isopropanol) containing Si-O network polymers. These above
reactions will continue with time even at room temperature and may
result in dense sol of larger network polymers. If this happens the SOG
will not be able to be applied uniformly on a substrate because of its
very high viscosity. In order to prevent these reactions the SOG should
be stored at low temperature (3oC is sufficient). Even though, the shelf
life of silicate SOG is limited to six months.
 Synonym: Orthosilicic
acid tetraethyl ester, TEOS, Tetraethoxysilane
Synonym: Orthosilicic
acid tetraethyl ester, TEOS, Tetraethoxysilane-
Linear Formula: Si(OC2H5)4, Molecular Weight: 208.33, CAS Number: 78-10-4
폴리에테르 실록산 공중합체를 더 포함하는 것이 바람직하다.(0.1~5.0 wt %)
유기 용매로 메틸이소부틸케톤을 사용할 수 있다.
트리에톡시실란 10.15g, 메틸이소부틸케톤 10g, 질산/물(0.0006% 수용액) 1.2g 및 물 1.2g을 50ml 플라스크에 칭량한후, 혼합하여 30 분간 교반하였다. 상기 반응 혼합물의 온도를 상온으로 냉각한 후, 이러한 반응 혼합물에 비스트리메톡시실릴에탄 1.85g 및 메틸이소부틸케톤 5.56g을 약 10 분에 걸쳐 투입하고, 3 시간 동안 다시 교반하였다. 이러한 과정을 통해 실시예 1의 절연막 형성용 조성물을 제조하였다.
실시예 2 : 본 발명의 절연막 형성용 조성물의 제조
실시예 1과 동일한 과정을 거쳐 조성물을 제조하고 나서, 이러한 조성물에 3.0 중량%(최종 제조되는 절연막 형성용 조성물의 총
중량 기준)의 폴리에테르 실록산 공중합체를 첨가하고 10 분 동안 교반하여, 실시예 2의 절연막 형성용 조성물을 제조
실시예 3 : 본 발명의 절연막 형성용 조성물의 제조
트리에톡시실란 10.15g, 메틸이소부틸케톤 10g, 질산/물(0.0006% 수용액) 1.2g 및 물 1.2g을 50ml
플라스크에 칭량한후, 혼합하여 30 분간 교반하였다. 상기 반응 혼합물의 온도를 상온으로 냉각한 후, 이러한 반응 혼합물에
비스트리메톡시실릴에탄 1.85g 및 메틸이소부틸케톤 2.3을 약 10 분에 걸쳐 투입하고, 3 시간 동안 다시 교반하였다. 이러한
과정을 통
해 실시예 3의 절연막 형성용 조성물을 제조하였다.
종래 기술
트리에톡시실란 11.27g, 메틸이소부틸케톤 15.56g, 질산/물(0.0006% 수용액) 1.2g 및 물 1.2g을 50ml
반응기에 넣고 4시간 동안 유지

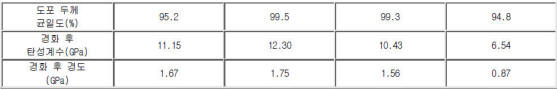
실리콘 화합물의 분자량 및 분자량 분포에 따라 많이 좌우된다.
분자량이 지나치게 크게 되는 경우: 젖음 특성이 떨어져서, 상 기 절연막 형성용 조성물이 종횡비가 크게 형성된 상기
배선 또는 전극 사이에 잘 스며들지 못하기 때문에, 절연막 내에 다수의 공동을 형성.
분자량 분포가 넓어질수록, 절연막 내의 입자와 입자 사이가 치밀하게 채워짐으로서, 평탄화가공 등에 의한 영향 자체가 최소화되어 절연막의 평탄화 특성이 향상되며, 절연막이 산 또는 염기 등에 대해서도 강한 저항성을 가지게 되고, 밀도가 큰 절연막이 형성되어 절연막의 기계적 물성이 향상된다
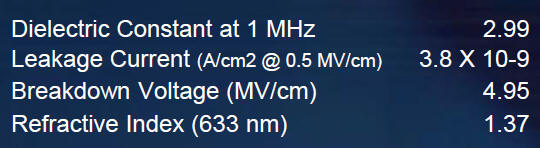
The micro porosity in silica is not removed entirely until 1000 oC, but it may already behave as an oxidation barrier or passivation coating at 600 oC. This ability of the micro-porous film to behave in many ways like the bulk oxide is an attractive property of the sol-gel approach to coating.
Silanol (Si-OH) compound에 CH3 기의 존재 유무에 따라 Silicate와 Siloxane 계로 나누어 지는데 CH3 기 가 없는 경우는 silicate 계이다.
The silicate SOG is formed from a condensation reaction of Si(OH)4 by losing water. When the film is fully cured, the film should form a strong Si-O network and contain no -OH but it has fairly significant shrinkage.
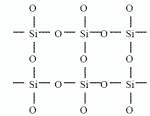
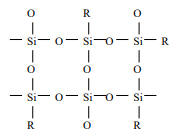
Siloxane type SOG는 Si 원자에 Silanol group과 Methyl group(-CH3)이 결합. Siloxane계는 Silicate계에 비해 점도가 커서 도포 후에 평탄도가 우수하고 Methyl group을 함유하고 있기 때문에 baking하는 과정에서 막의 수축을 막을 수 있으며 이로 인한 stress도 작아 막의 Crack의 저항성이 크다는 장점,
contains –CH3 or –C2H5
types of organic dopants. the organic group modifies the Si-O
network which lowers the film stress to 200 MPa, typically. The
viscosity and molecular weight are slightly higher than that of the
silicate SOG. The film is thicker than that of the silicate SOG using
the
same spin speed and its planarity is better
It has been observed that some of the siloxane SOG films pick up atmospheric water and this may become a reliability issue or cause extensive processing constraints.
It can be seen from the FTIR spectra before and after 400 oC baking that Si-OR and Si-R (R stand for organic groups) peaks disappear and Si-OH peaks show up.
The - OH content not only reduces the device performance (due to high dielectric constant) but also creates reliability issues.
Increasing the organic content will further reduce the film stress and improve the crack resistance and planarity.
Siloxane 계가 Film내 Carbon이 O2와 반응하는 이유로 Silicate 계에 비해 수축이 크다. 또한 420℃/30분 Anneal 후에 H2O와 CH3와 같은 유기물이 발견되지 않아 최소한 420℃ 정도의 Curing이 필요하다.
Siloxane 계 Silicate 계
N2 분위기 10.7% 20.5%
O2 분위기 14.4% 19.9%





·장점 : Liquid source의 Coating 형태로 평탕화 효과가 뛰어나고, 비용이 저렴.
·단점 : Curing시 H2O의 Outgassing으로 인한 Crack 발생 및 Film내에 존재하는
Carbon에 의한 Polymer 형성 및 Via 저항 상승.
| |
OH - Si - OH + O = X - OR + Solvent SOLUTION
| |
OH OH
| | |
O - Si - O - Si - O + O = X - OR POROUS
| | |
OH O OH (After Spinning ∼ 150℃)
| | ∥ |
O - Si - O - Si - O - X - O - Si - O - GLASSY
| | | |
O O O O (Heated at 250℃ ∼ 350℃)
| | |
- Si - OR - Si -
| |
| | ∥ |
O - Si - O - Si - O - X - O - Si - O - INORGANIC
| | | |
O O O O (Heated at 350℃ ∼ 600℃)
| | | |
O - Si - O - Si - O - X - O - Si - O -
| | | |
O O O O
| | | |
O - Si - O - X - O - Si -
| ∥
O O
네패스 특허
Cospeen-1225G is methyl-siloxane-based organic spin-on-glass coating solution
• Coating thickness 2300 Å - 4000 Å per coat
• Bake temperature / time 80oC / 2 min - 230oC / 4 min
• Cure temperature / time 450oC / 30 min in N2